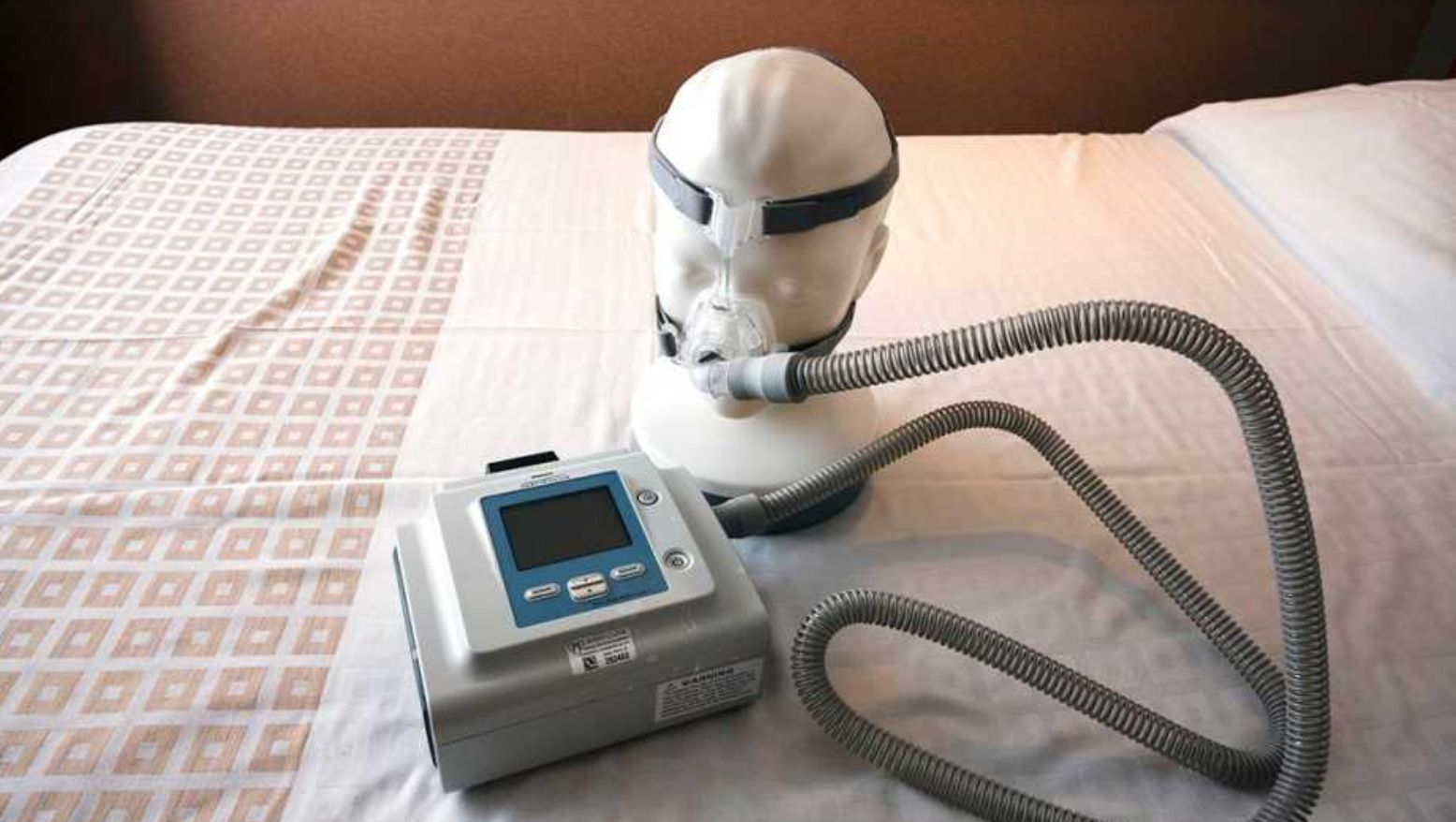
philips recalled 5.5 million CPAP machines and 5 years later it's still not over the polyurethane foam inside degraded into toxic particles patients inhaled them while sleeping FDA received 69,000 ad…

LinkedIn Content Strategy & Writing Style
Hardware development for next-gen medical devices | Author of Hardware Bible: Build a Medical Device from Scratch
1 person tracking this creator on Viral Brain
Lisa Voronkova positions herself as the pragmatic voice of authority for medtech founders, bridging the gap between ambitious hardware engineering and the brutal realities of regulatory compliance. Her content strategy centers on a "reality check" value proposition, using high-stakes case studies like Philips recalls or Neuralink data to expose the hidden bottlenecks in sterilization, tariffs, and AI legislation. She is notable for her ability to translate dense technical risks into clear financial trade-offs, often arguing that rigorous design controls are a profit-protection strategy rather than a bureaucratic hurdle. By intersecting deep engineering expertise with strategic business consulting, she successfully frames her services as the essential safeguard against the $50 million recalls that sink under-prepared startups.
14.8K
12.3K
122
—
5.1
94
2
philips recalled 5.5 million CPAP machines and 5 years later it's still not over the polyurethane foam inside degraded into toxic particles patients inhaled them while sleeping FDA received 69,000 ad…

Boston Scientific just got FDA clearance for their second PFA catheter Farapoint approved January 14th adds to Farawave portfolio pulsed field ablation is transforming AFib treatment energy pulses…

new Class III device approved: prescription at-home neuromodulation for treatment-resistant depression Neurolief's Proliv™Rx got PMA approval January 12th read that again: PMA approval for brain neu…

we saved a client $200K by killing their favorite feature they hated us for a week thanked us for a year startup building a wearable cardiac monitor. they wanted continuous ECG, SpO2, temperature, a…

neuralink has 21 patients with brain implants controlling computers with their thoughts playing video games, designing in CAD, making phone calls this isn't science fiction anymore the 2026 data: b…

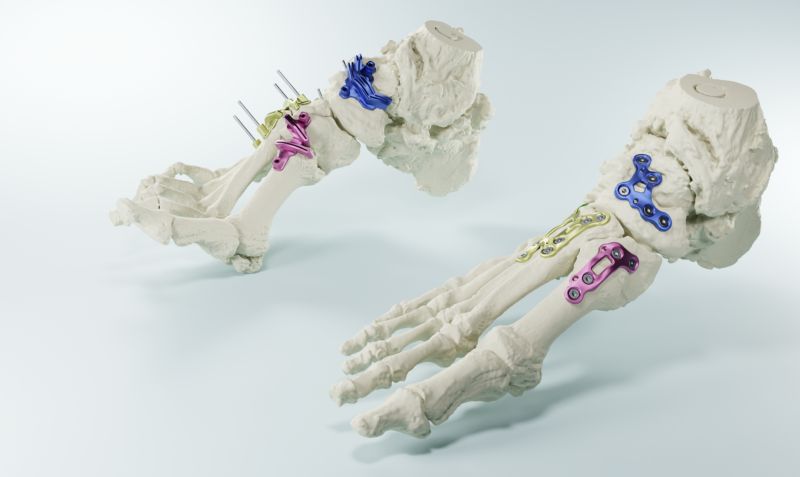
MedCAD just got FDA clearance for patient-specific 3D-printed titanium plates custom foot and ankle implants designed from each patient's CT scan sounds revolutionary here's the problem: patient-sp…
5.1 posts/week
Posts / Week
1.6 days
Days Between Posts
2
Total Posts Analyzed
HIGH
Posting Frequency
121.75%
Avg Engagement Rate
STABLE
Performance Trend
215
Avg Length (Words)
HIGH
Depth Level
ADVANCED
Expertise Level
0.83/10
Uniqueness Score
NO
Question Usage
0.1%
Response Rate
Writing style breakdown
<start of post>
the hidden cost of "regulatory shortcuts" is exactly $42 million
at least for one startup we met last quarter
they had a class II cardiovascular implant
investors were pushing for a Q4 launch
so they "optimized" their clinical evaluation report by reusing data from a predicate device that wasn't a 100% match
they saved $150K on clinical consultants
they saved 6 months of trial time
then the notified body did a deep dive
the gap in clinical evidence was flagged immediately
not only was their submission rejected, but they were put under a "high-scrutiny" review for all subsequent filings
that 6-month "saving" turned into an 18-month delay
investors pulled out during the bridge round
the company folded in january
this isn't a regulatory problem
it's a math problem
$150K in savings → $42M in lost enterprise value
we see this every time a founder says "we'll fix the documentation in v2."
in medtech, there is no v2 if v1 doesn't clear the bar.
quality is not a department. it's a survival strategy.
if you're building hardware and want my team to handle the engineering while you focus on business - link in bio
repost if you've seen a project killed by "saving time" on regulatory
follow me at Lisa Voronkova for more on the real risks of medtech development
<end of post>
Sign in to unlock the full writing analysis
Nail your LinkedIn strategy with ViralBrain.
Analyze and write in Lisa Voronkova's style. Grow your LinkedIn to the next level.